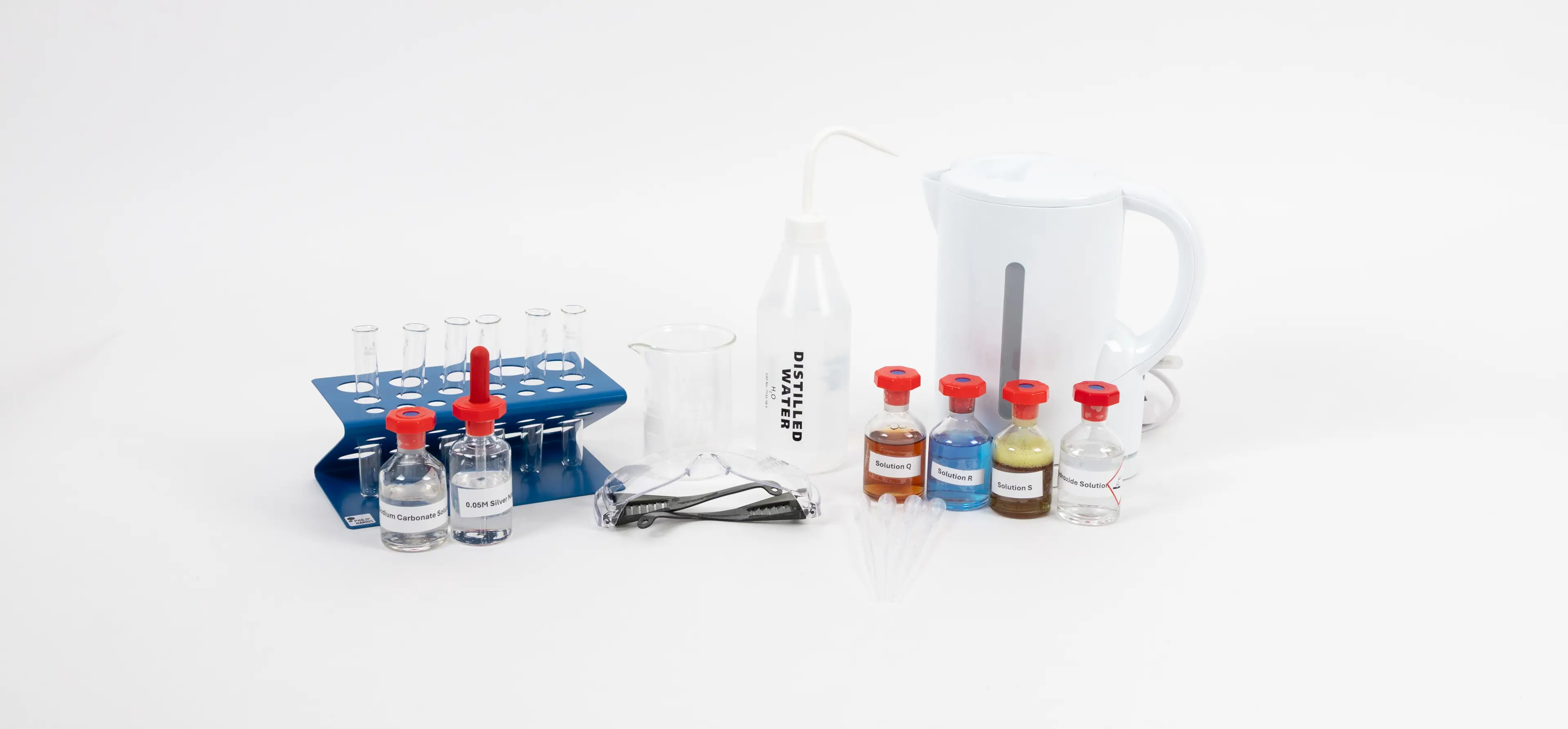
Identifying transition metal ions A Level Chemistry
Carry out simple test-tube reactions to identify transition metal ions in aqueous solution by the colour in a precipitate.
Subject: Chemistry | Level: A Level |
You will need:
Method
Test 1(a):
Place 10 drops of solution Q in a test tube
Add Sodium Hydroxide solution, dropwise with gentle shaking, until in excess
Do not discard this mixture
Repeat this test with solution R and then solution S
Record your results in a suitable table
Test 1(b):
Half fill a 250ml beaker with freshly boiled water
Allow the four test tubes containing the mixtures from test 1 (a) to stand in the beaker of hot water for about 10 minutes
Record your results in a suitable table
Test 2:
Place 10 drops of Sodium Carbonate solution in a test tube
Add 10 drops of solution Q and shake the mixture gently
Repeat this procedure with solution R and then solution S
Record your results in a suitable table
Test 3:
Place 10 drops of solution Q in a test tube
Add 10 drops of Silver Nitrate solution and shake the mixture gently
Repeat this procedure with solution R and then solution S
Allow the four test tubes to stand for 10 minutes
Record your results in a suitable table
Theory:
Transitions metals absorb some frequencies of visible light as electrons jump to higher orbitals. Meaning the complex ions formed are usually coloured.
Transition metals form insoluble metal hydroxides when they react with Sodium Hydroxide, we can identify the metal from the colour of the precipitate.
Fe3+(aq) + 30H-(aq) → Fe(OH)3(s)
Iron (III) will from a yellow/ brown precipitate.
Cu2+(aq) + 20H-(aq) → Cu(OH)2(s)
Copper (II) will produce a blue precipitate
Fe2+(aq) + 20H-(aq) → Fe(OH)2
Iron (II) will produce a green precipitate












