
Measuring enthalpy change A Level Chemistry
Students measure enthalpy changes using calorimetry to measure the measuring the temperature change of a known mass of water or solution. The heat energy absorbed or released, q, is calculated using the formula q = mcΔT (where m is the mass, c is the specific heat capacity, and ΔT is the temperature change). This heat change is then used to calculate the molar enthalpy change (ΔH) for the reaction.
Subject: Chemistry | Level: A Level |
You will need:
Method
Experiment One – Collecting data for the determination of ΔH1
Weigh approximately 4g of anhydrous Copper (II) Sulfate in a dry stoppered weighing bottle
Record the precise mass of Copper (II) Sulfate
Construct a suitable table to allow results to be recorded every minute over a 15-minute period
Using a 25ml measuring cylinder, place 25ml of distilled water into the insulated cup and record the initial temperature (t = o)
Start the timer and record the temperature every minute, ensuring you are stirring the liquid continuously
At the fourth minute add the anhydrous Copper (II) Sulfate rapidly to the water and continue to stir. But do not take the temperature
At the fifth minute record the temperature and do this every minute up to 15 minutes
Plot a graph with temperature on the y axis against time
Draw 2 separate lines of best fit. Line one which joins the points before the addition. Line two which joins the points after the addition
Extrapolate both lines to the point of 4 mins
Use your graph to determine the temperature change at the fourth minute
Experiment 2 – Collecting data for the determination of ΔH2
Weight out approximately 6.2g of Copper (II) Sulfate-5-water into a dry stoppered weighing bottle
Construct a suitable table of results to allow the recording of the temperature at 1-minute intervals up to a total of 15 minutes
Using a 25ml measuring cylinder, measure 24mls of distilled water into an insulated cup
Record the temperature of the water and start the stopwatch
Take the temperature every minute whilst continuously stirring the water
At minute 4, add the Copper (II) Sulfate-5-water quickly and continue to stir
At the fifth minute, record the temperature and do this every minute up to minute 15
Plot a graph with the temperature on the y axis against time
Draw 2 separate lines of best fit. Line one joining the points up to 4 minutes. Line 2 which joins the points after the addition
Extrapolate both lines to minute 4
Use your graph to determine the temperature change at the fourth minute
Analysing the data:
Heat change = mass x specific heat capacity x temp change
Heat change = M x C x ΔT
Note : Specific heat capacity (SHC) of cup negligible compared to the mass of water
Note: SHC of the aqueous solution can be considered to be the same as water
For both experiments you will need the mass of water in (g). Density of water 1g/cm³ the mass is therefore the same as the volume used, which in both cases was 25g
You will need the temp change in K from the graph
SHC for water – 4.18 JK-1 g-1
Value obtained for heat change will be in J. convert to KJ by dividing 1000 Calculate the enthalpy changes for ΔH1₁ and ΔH2₂ in KJ/Mol, using the masses for the solids Use two values of ΔH1 and ΔH2 and apply Hess’s Law and calculate ΔH3 for the hydration of CuSO4
Theory:
Hess’s Law:
Enthalpy change for a chemical reaction is independent of the route taken
This means that the enthalpy change for the overall process will be the same independent of the steps taken
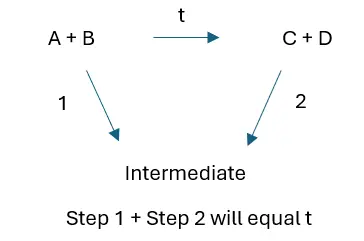
Take the following reaction –
A + B → C + D
The enthalpy change to form products C + D directly will be the same as the sum of the enthalpy changes for the production of C + D by an undirect route, where there will be intermediate products formed
Technician tips
Keep the stock of solid anhydrous Copper (II) Sulfate in a closed container during weighing
The crystals already contain water so the total amount of water will be the same as experiment one













