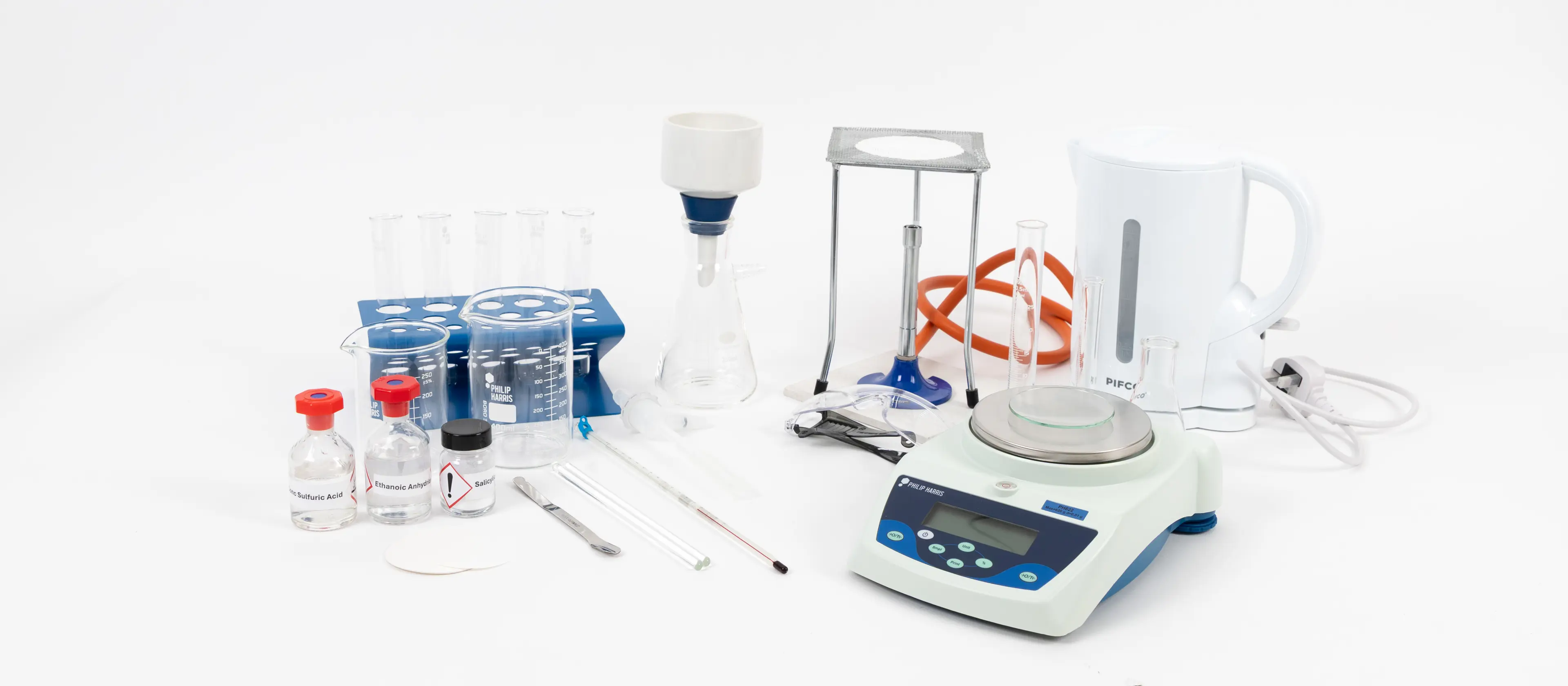
Preparation and purity test of an organic solid A Level Chemistry
Students prepare and test the purity of an organic solid by measuring its melting point. Pure solids will melt at a precise temperature, whereas impure solids melt at a range of temperatures.
Subject: Chemistry | Level: A Level |
You will need:
Method
Part 1 Method: Preparation
Weigh out approximately 6g of Salicylic Acid directly into a 100ml conical flask
Record the mass of salicylic acid used
Using a 10ml measuring cylinder, add 10ml of Ethanoic Anhydride to the flask and swirl the contents
Carefully add 5 drops of concentrated sulfuric acid to the flask and swirl the mixture in the flask for a few minutes to ensure thorough mixing
Warm the flask for about 20 minutes in a 400ml beaker of hot water at approximately 60⁰C.
Note: The temperature in the flask should not be allowed to rise above 65⁰CAllow the flask to cool and pour its contents into 75ml of water in a beaker, stirring well to precipitate the solid
Filter off the aspirin under reduced pressure, avoiding skin contact
Collect the crude aspirin on a double thickness of filter paper and allow it to dry
Part 2 Method: Purification
Using a 25ml measuring cylinder, measure out 15ml of Ethanol into a boiling tube
Prepare a beaker half filled with hot water at a temperature of approximately 75⁰C
Note: The safest way to do this is by using a kettle of boiling water and add water from the kettle to cold water in the beaker until the temperature is approximately 75⁰C
Note: The boiling point of ethanol is 78⁰C and the temperature of the water in the beaker should not be allowed to go above thisUse a spatula to add the crude Aspirin, prepared in part one in to the boiling tube and place the tube in the beaker of hot water. Do not scrape the filter paper.
Stir the contents of the boiling tube until all of the Aspirin dissolves into the Ethanol
Pour the hot solution containing dissolved aspirin into approximately 40ml of water in a 100ml conical flask. If a solid separates at this stage, gently warm the contents of the flask in the water bath until the solid is completely dissolved into solution. You should avoid prolonged heating, since this will decompose the aspirin
Allow the conical flask to cool slowly and white needles of aspirin should separate
If no crystals have formed after the solution has cooled to room temperature, you may need to scratch the insides of the flask with a glass stirring rod to obtain crystals. Cool the whole mixture in an ice bath
Filter off the purified solid under reduced pressure and allow it to dry on filter paper
Record the mass of the dry purified solid
Analysing the effectiveness of this method of preparation of aspirin:
Calculate the theoretical yield of aspirin which should be formed from 6g of salicylic acid
Expected yield for Aspirin:
Balanced equation:
C7H6O3 + C4H6O3 → C9H6O4 + C9H8O4 + CH3COOH
Relative formula Mass:
Salicylic Acid: 7 x 12 (C) = 84
6 x 1 (H) = 6
3 x 16 (O) = 48
= 138
Ethanoic Anhydride: 4 x 12 (C) = 48
6 x 1 (H) = 6
3 x 16 (O) = 48
= 102
Aspirin: 9 x 12 (C) = 108
8 x 1 (H) = 8
4 x 16 (O) = 64
= 180
Calculate the moles of Salicylic Acid:
Moles = Mass/ Molar Mass Moles= 2g /138g/mol = 0.014mol
Calculate the moles of Ethanoic Anhydride:
Work the amount of Ethanoic Anhydride out in (g) using the density 1.08g/ml
Amount in (g) = amount in ml density 10ml 1.08g/ml = 10.8g
Moles = Mass / Molar Mass Moles= 10.8/ 102 = 0.11mol
Work out the limiting reactant: In this equation the mole ratio is 1:1.
We have fewer moles of Salicylic Acid so this is our limiting reactant.
Calculate the moles of Aspirin:
Using the balanced equation 1 mol of Salicylic Acid makes 1 mol of Aspirin
0.014mol of Salicylic Acid = 0.014mol of Aspirin.
Theoretical Yield of Aspirin = moles x molar mass Theoretical Yield = 0.014 * 180 = 2.52g
Calculate the moles of Salicylic Acid:
Calculate the percentage yield of aspirin from your experiment and comment on the reasons for the losses that have occurred during the preparation and the purification of the solid
Test the purity of an organic solid by measuring its melting point:
The purity of an organic solid can be determined in part by measuring its melting point and comparing the value with the known Data Book value of the melting point for that compound.
A pure dry solid will melt at a precise temperature whereas an impure solid will melt over a range of temperatures which are lower than the melting point of the pure solid
When melting occurs, the solid should collapse into a liquid without any change in temperature and the way in which it occurs can give a clue to the purity of the solid
Several measurements should be taken to obtain a reliable value for the melting point.
Method
Powder a sample of the organic solid by crushing it gently with a spatula onto the surface of a filter paper
Fill three melting point tubes with the organic solid to a depth of approximately 0.5cm
Set up the melting point apparatus provided and mount one of the melting point tubes ready for taking a measurement
Heat the apparatus gently and observe the temperature at which the solid collapse into a liquid. The melting point will be in the range 100⁰C to 200⁰C
Allow the melting point apparatus to cool and repeat the measurement of the melting point of the solid with the other two samples. Use the first reading as an approximate value, then the heating of the other two samples can be done much more slowly as the temperature reaches the approximate value.
Use the 3 measurements that you have recorded to work out the mean value.
Compare the data book value for the true melting point of the solid and compare to your sample.




























