
Test for alcohol, aldehyde, alkene and carboxylic acid A Level Chemistry
This experiment is divided into five parts. During each part of this experiment, it is important that students present all of their observations in a neat table. The skill of being able to present clearly organised records of observations is an important skill, and students are expected to demonstrate this skill as part of carrying out this set of experiments
Subject: Chemistry | Level: A Level |
You will need:
Method
Part 1 – Testing for Alcohols
Add 1ml of Ethanol to a clean, dry test tube
Very carefully add a small piece of metallic Sodium
Record your observations in a suitable table
In the presence of a functional alcohol group the Sodium will react, the student will observe the metal Sodium fizzing with bubbles of Hydrogen gas being released
2C2H5OH + 2Na → CH3CH2Ona + H2
2(C2H5ONa)
Part 2 – Testing for Aldehyde using Fehling’s solution
Add 2mls of Fehling's solution A and Fehling's solution B into a clean, dry test tube, and shake to mix. The solution should be a clear dark blue solution
To a separate test tube add 1ml of Sodium Carbonate solution and some Anti bumping granules, add 5 drops of the prepared solution from step one
To the same test tube add 1ml of Ethanal
Warm the test tube gently for around 2 minutes in a beaker that is half filled with hot water
Gradually bring the beaker of water to boiling and maintain the water bath at this temperature for a few minutes (CLEAPSS advice: stand test tube in a beaker of just boiled water from a kettle, instead of using a Bunsen Burner)
Using a test tube holder remove the test tube from the water bath. Allow the solution to cool for several minutes and observe and record the results in a suitable table.
A positive test will give a brick red precipitate
Fehling’s reagent is Copper (II) complex made by adding two solutions together:
Fehling’s solution A – contains Copper (II) Sulfate Fehling’s
solution B – contains Sodium Hydroxide and Potassium Sodium Tartrate
When the mixture is warmed with an aldehyde, the brick red precipitate is Copper (I) Oxide which is formed as the Copper (II) is reduced to Copper (I)
CH3CHO+ 2Cu2+ + SOH- → CH3COO- + CU2O + 3H20
(Fehling’s A) (Fehling’s B) (insoluble ppt)
Part 3 – Test for an Alkene (a test for unsaturation)
Add 2 drops of Cyclohexene to a test tube
Add 1ml of Bromine water to the same test tube and shake the contents to mix well
Observe and record results in a suitable table
A positive result will see the Bromine water change from the orange/red colour to colourless
An unsaturated organic molecule contains a C=C double bond. When Bromine water is added the double bond opens up and a dibromide is formed
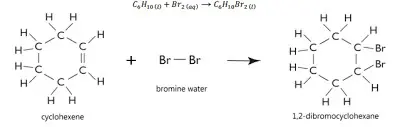
C6H10 + Br2 → C6H10Br2
Part 4 – Testing for Carboxylic Acid
Add 2 drops of Cyclohexene to a test tube
Place a spatula of Sodium Hydrogen Carbonate in a boiling tube
Add 2mls of dilute Ethanoic Acid to the same boiling tube
Observe and record your results
Students will observe bubbles in the solution as carbon dioxide is formed as the Ethanoic Acid reacts with the Sodium Hydrogen Carbonate
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Part 5 – Testing for a Haloalkane
To a clean, dry test tube add 1ml of Sodium Hydroxide solution
Using a pipette add 5 drops of 1-Bromobutane
Warm the test tube by placing in a beaker filled with warm water (around 60⁰C) and leave for a few minutes
Acidify the solution by adding 2mls of dilute Nitric Acid, followed by 1ml of Silver Nitrate solution
Observe and record your results in a suitable table
With a positive result students will observe the formation of a very pale cream precipitate when the Sodium Hydroxide is added, the halogen (Br) atom from the Haloalkane is displaced as a Halide ion
C4H9Br + NaOH → C4H9OH + NaBr
As an ionic equation:
C4H9Br + OH- → C4H9OH + Br-
Acidifying this reaction ensures that any unreacted Hydroxide ions do not react with the Silver ions to give a confusing result with precipitate
Addition of Silver Nitrate allows the Halogen ion (Br¯) to react with the Silver ion to form an insoluble Silver Bromide precipitate
AgNO3 + NaBr → AgBr + NaNO3
Technician tips
Unless otherwise stated it is recommended to use concentrations of 2M for solutions
It is important to use clean, dry test tubes























