
Distillation of a product from a reaction A Level Chemistry
Students are required to distill a product from a reaction by heating a mixture in a round-bottom flask to vaporize the desired product, which has a lower boiling point than other components.
Subject: Chemistry | Level: A Level |
You will need:
Method
This experiment needs to be carried out in a fume cupboard
Weigh a 50ml round bottomed flask, pour 20ml of Cyclohexanol into the flask, and reweigh. Record the mass of Cyclohexanol
Using a plastic graduated dropping pipette, very carefully and with frequent shaking, add to the flask 8mls of Concentrated Phosphoric Acid
Add a few anti bumping granules to the flask and assemble the distillation equipment as below
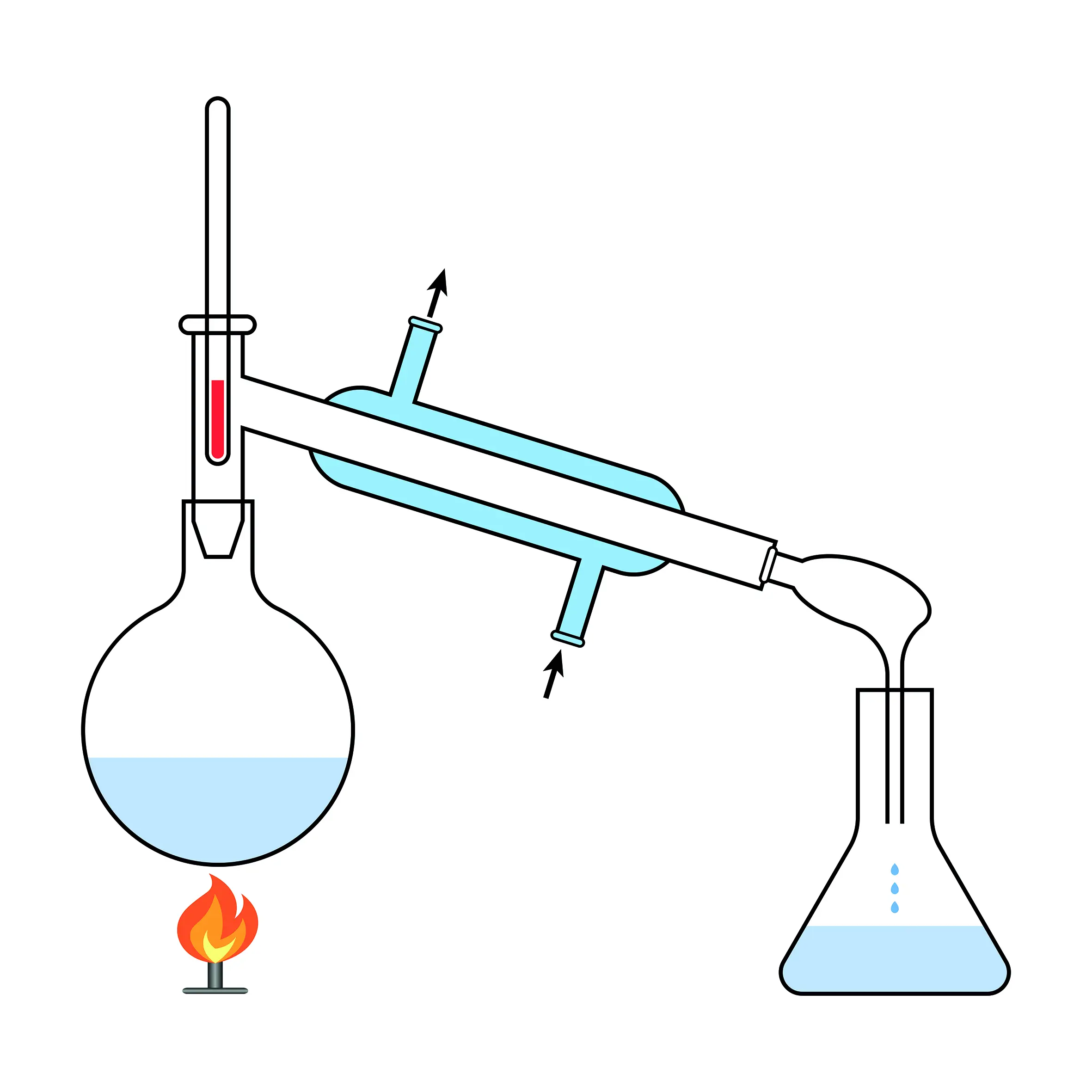
Heat the flask gently, distilling any liquid which boils below 100⁰C
Pour the distillate into a separating funnel and add 50mls of saturated Sodium Chloride solution, shake the mixture and allow the 2 layers to separate
Once separated run off the lower layer into a beaker
Transfer the upper layer, which contains the crude cyclohexene into a small conical flask
Transfer a few lumps of anhydrous Calcium Chloride to a weighing boat and add to the crude Cyclohexene to remove water
Stopper the flask, shake the contents then allow this to stand until the liquid becomes clear
Decant the liquid into a clean, dry weighed beaker
Reweigh the filled beaker, calculate the mass of the dry Cyclohexene produced and determine the percentage yield of your product
Test the distillate to confirm it contains an alkene
Test for an alkene
Add approximately 1ml of your distillate into a clean, dry test tube
Add equal amount of acidified Potassium Manganate (VII) solution, shake the contents of the test tube and record your observations
Calculating percentage yield
Percentage yield = mass obtained / expected mass x 100
Expected yield for dehydration of Cyclohexanol
Balanced equation
C6H11OH → C6H10 + H2O
Relative formula mass of both substances
Cyclohexanol 6 x 12 (C) = 72
1 x 12 (H) = 12
1 x 16 (O) = 16
= 100
Cyclohexene 6 x 12 (C) = 72
1 x 12 (H) = 12
1 x 16 (O) = 16
= 100
100g of Cyclohexanol should produce 82g of Cyclohexene
Reacting masses :
100/100=1g Cyclohexanol produces 82/100 g Cyclohexene
If we presume that we used 20g of Cyclohexanol
The expected yield should be –
82/100 x 20g = 16.4g
Testing for alkene
Acidified Potassium Manganate (VII) which is a purple solution will become colourless in the presence of an alkene
The Potassium Permanganate is a strong oxidising agent and will break the Carbon-Carbon double bond. As this reaction proceeds, the purple colour of the Potassium Permanganate will turn colourless.
Technician tips
This experiment needs to be carried out in a fume cupboard


























